Laboratory of Cell Biology
Laboratory overview
(for more information, see also our webpages at www.cell-biology.cz)
Our primary mission is to identify and understand the molecular modules that regulate plant cell polarity and morphogenesis, focusing on those operating at the intersection of the secretory pathway, plasma membrane lipids, and the actin cytoskeleton. We divide our attention between two main processes that give plant cells their shape: oriented cell division and differential cell growth, and we focus on intracellular molecular mechanisms driving cellular morphogenesis, such as exocytosis.

Our research interests
The orchestration of cellular processes in eukaryotes hinges on transporting extracellular matrix, membrane lipids, and proteins to the cell surface, predominantly achieved through exocytotic vesicles. The culmination of the secretory pathway is exocytosis, a finely tuned process involving the tethering, docking, and fusion of secretory vesicles with the plasma membrane, ultimately culminating in cargo release.
At the core of our investigations lies the exocyst, a conserved octameric protein complex that plays a central role in tethering secretory vesicles to the plasma membrane, thus serving as a pivotal exocytosis regulator. Our research efforts are centred around dissecting the functional intricacies of the exocyst across a diverse spectrum of plant species, ranging from angiosperms Arabidopsis thaliana and Nicotiana tabacum to bryophytes Physcomitrium patens and Marchantia polymorpha, as well as the streptophyte alga Klebsormidium nitens. An important aspect of our research is exploring the role of vesicular traffic, specifically the exocyst, in plant reaction to abiotic stresses and in plant-microbe interactions.
Concurrently, our research extends into deciphering the roles of negatively charged phospholipids in establishing and maintaining plant cell polarity. Although quantitatively minor, these anionic lipids are pivotal in governing membrane characteristics, charge, curvature, signalling, and protein recruitment. The collaborative regulatory role of these phospholipids in orchestrating vesicular traffic at the plasma membrane is of particular interest. In this context, we also examine the lipid kinases and phospholipases that catalyze the production of anionic lipid phosphatidic acid at the plasma membrane.
Significant results 2021-2023
Molecular insight into the function and evolution of plant exocyst complex
We have uncovered the modular structure of the exocyst complex, pinpointing the role of the EXO70A1 subunit in tethering the plant exocyst to the lateral plasma membrane in roots. This binding is mediated through interactions with an array of anionic phospholipids. Through a multifaceted approach encompassing biochemical, genetic, microscopic, and computational techniques, we have shown the role of phosphatidylinositol 4-phosphate and phosphatidic acid in controlling exocyst binding to the plant plasma membrane (Fig. 1). This study also emphasizes the pivotal role of membrane charge in governing interactions between peripheral proteins and membranes (Synek, Pleskot, Sekereš et al. 2021).
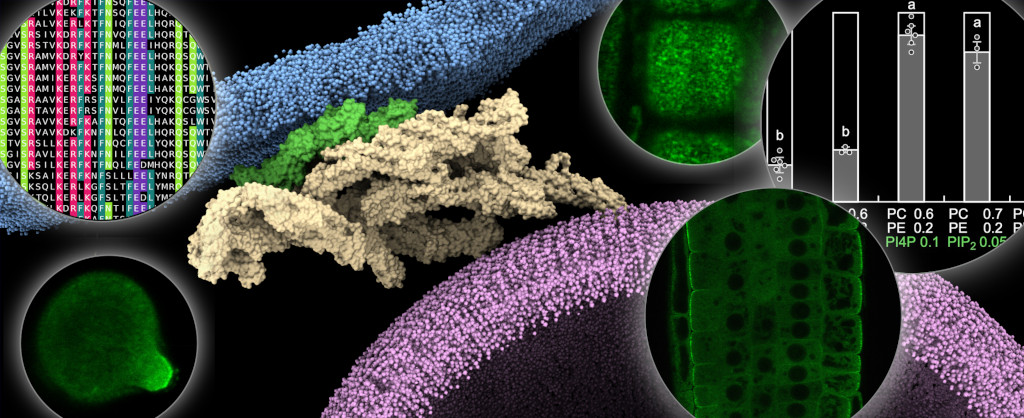
Fig. 1: Model suggesting how Arabidopsis exocyst complex tethers a secretory vesicle to the plasma membrane via the isoform EXO70A1 and multiple anionic phospholipids. The insets showcase the multifaceted workflow of our lab, which combines state-of-art microscopical, computational, biochemical, and in planta approaches. Inspired by the results published in (Synek, Pleskot, Sekereš et al. 2021).
We elucidated the evolutionary history of the land plant exocyst complex, revealing a multistep pattern of evolution of distinct exocyst subunits. We have dissected the evolution of the EXO70 subunit, revealing its division into three well-established subfamilies, each exhibiting unique functional attributes. Through cross-complementation assays, we have highlighted the deep conservation of the canonical EXO70.1 subfamily and the independent evolution of non-canonical EXO70.2 and EXO70.3 subfamilies (Haluška, Janková-Drdová et al., submitted). Additionally, our investigations into the SEC15a and SEC15b exocyst subunit isoforms have illuminated their preferred roles in the male gametophyte and sporophyte, respectively, and revealed a surprising acquisition of novel function for SEC15a in the sporophyte (Batystová et al. 2022). Our functional study of the exocyst in the moss Physcomitrium patens has provided insight into the significance of SEC6 exocyst subunit in cell division and organization of cell assemblies. Our observations document a role for the exocyst complex in the transition from simple filamentous structures to complex morphological arrangements in plant organs (Fig. 2) (Brejšková et al. 2021).

Fig. 2: Disruption of the exocyst complex in the moss Physcomitroium patens leads to severe developmental defects. Adapted from (Brejšková et al. 2021).
Role of vesicular traffic in plant-microbe interactions
In the reaction to pathogenic fungi, plants employ intensive exocytosis to deposit cell wall reinforcements (papillae or encasements), which block the spread of infection throughout the plant. We identified a vital role for the exocyst complex containing subunit EXO70B2 in the papillae membrane domains important for callose deposition and encasement formation. Our results have highlighted the importance of EXO70B2-containing exocyst in delivering the key SNARE protein SYP121 to the papillae membrane domains, offering new insights into mechanisms of penetration resistance. (Ortmannová et al. 2022). We found that a closely-related exocyst subunit EXO70B1 plays a vital role in responses to various abiotic stresses, operating at both endomembranes and plasma membrane in a complex cooperation/competition mode with EXO70B2 (Drs et al. in preparation).
Our research shed new light on the enigmatic cellular processing, trafficking, localization, secretion, and function of the pathogenesis-related 1 (PR1) protein. By generating a spectrum of tagged Arabidopsis PR1 variants, we have provided insights into the dynamic modulation of defence responses and immune pathways through PR1 protein processing. This intricate interplay is dependent on cellular localization and plant age, highlighting the multifaceted nature of this crucial plant defence protein (Pečenková et al. 2022).
To unravel the interplay between cell morphogenesis and biotic stress responses, we began to investigate how the immunity elicitor chitosan impacts the polar growth of plant cells. We found that chitosan triggers root hair callose deposition and growth inhibition. Our findings revealed the deep conservation of this strategy in analogous root hair-like structures in lycopods and bryophytes, underpinning the significance of this response in mild biotic stress situations. (Drs et al., submitted).
Lipid signaling at the plant plasma membrane
Pollen tubes require a tightly regulated pectin secretion mechanism to maintain the cell wall plasticity required for polar tip growth. Phosphoinositides and phosphatidic acid participate in this regulation at the apical plasma membrane, but the processes regulating their production remain unclear. In collaboration with Till Ischebeck's group at the University of Göttingen, Germany, we have shown that diacylglycerol kinase 5 plays a significant role in the regulation of pectin secretion in pollen tubes growing at the apex (Fig. 3) (Scholz, Pejchar et al. 2022).
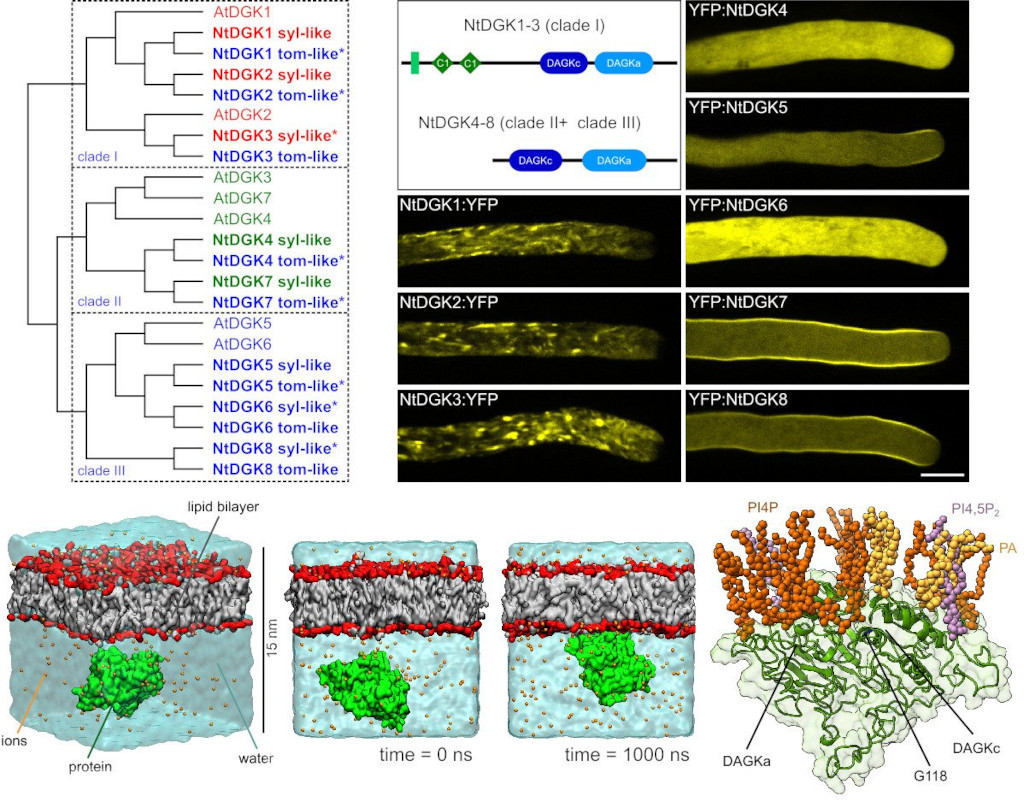
Fig. 3: Tobacco diacylglycerol kinase isoforms show distinct localization patterns in pollen tubes and bind to the plasma membrane via phosphoinositides. Left, phylogeny of Arabidopsis and Nicotiana tabacum diacylglycerol kinase (DGK) isoforms and their distribution into three clades. Right, domain distribution and localization of YFP-tagged NtDGK1–8 in actively growing pollen tubes. Lower panel illustrates the molecular dynamics simulations showing mechanistic details of the NtDGK5–membrane interaction. Scale bar, 10 μm. Adapted from (Scholz, Pejchar et al. 2022).
In two extensive collaborative studies led by Daniel Van Damme (PSB VIB Ghent, Belgium) and Roman Pleskot (Laboratory of Integrative Structural Biology, IEB), our group joined forces to unravel the role of anionic lipids in the regulation of plant clathrin-mediated endocytosis, namely the structure and regulation of the TPLATE complex (TPC). We focused on the lipid-binding properties of several TPC subunits, showing distinct but complementary roles of anionic lipids in the recruitment of TPC to the plasma membrane (Yperman et al., 2021a, 2021b). Recently, we revealed that anionic lipids play a role in the biogenesis of biomolecular condensates of specific TPC subunits (Dragwidge et al., submitted).
As membrane lipids are important players in plant responses to various stresses, we investigated the involvement of lipid signalling in flagellin perception, which is a keystone of pattern-triggered immunity in plants. In collaboration with the Laboratory of Pathological Plant Physiology and the group of Eric Ruelland (CNRS, Compiègne, France), we found that flagellin-derived peptide flg22 caused rapid and transient changes in Arabidopsis lipid dynamics. We identified diacylglycerol kinase 5 (DGK5), the enzyme producing phosphatidic acid, as the responsible gene. Importantly, dgk5.1 mutant plants produced less phosphatidic acid in response to flg22 and showed impaired resistance. The enzymatic activity of plasma membrane-localized DGK5 is thus vital for flagellin signalling and early immune responses in plant–microbe interactions (Kalachova, Škrabálková et al., 2022).

Fig. 4: Collection of cover pages published between 2021-2203, which highlights the outcomes of our work (Batystová et al. 2022, Synek et al. 2021, Markovic et al. 2020).
Workers
head of the laboratorysenior scientist
- Ing. Martin Potocký Ph.D.
deputy head of the laboratoryscientist
- Tamara Pečenková Ph.D.
senior scientist
- RNDr. Viktor Žárský CSc.
scientist
- Mgr. Edita Drdová
- RNDr. Michal Hála Ph.D.
- Ing. Přemysl Pejchar Ph.D.
- Mgr. Lukáš Synek PhD.
scientific assistant
- Mgr. Lucie Brejšková PhD.
- Ing. Andrea Potocká Ph.D.
- Mgr. Hana Soukupová Ph.D.
postdoc
- Mgr. Jitka Ortmannová PhD.
- MSc. Ankush Saddhe PhD
postgraduate student
- Mgr. Klára Batystová
- Msc. Laiju Chandran Ka...
- Bc Matěj Drs
- Samuel Haluška
- Ing. Ondřej Novotný
- Mgr. Eliška Škrabálková
technician
- Bc. Jana Šťovíčková
Research projects
- All roads lead to ROS: spatio-temporal regulation of pollen NADPH oxidases (Czech Science Foundation - GAČR project GA , GA ČR , Přemysl Pejchar
- FASS/TON2 functions in moss cell morphogenesis and phylogenetic insights into the TTP complex evolution , GA ČR , Viktor Žárský
- The plasticity of protein-lipid interfaces in plant membranes perceived through evolutionary lenses , GA ČR , Martin Potocký
- Good-Cop/Bad-Cop: Distinct roles of anionic phospholipids in plant endocytosis , GA ČR , Martin Potocký
- SIGNALS - The role of anionic phospholipids in the regulation of endocytosis in tip-growing plant cells , MŠMT , Martin Potocký
- Control of plant exocyst function by protein phosphorylation in root hairs and pollen tubes - role of unconventional exocyst complex subunits EXO70C , GA ČR , Viktor Žárský
- Multifaceted analysis of diacylglycerol kinase family in plants , GA ČR , Přemysl Pejchar
- Plant exocyst complex function in autophagy-related membrane transport , GA ČR , Tamara Pečenková
- Multiscale analysis of signalling phospholipids and their interaction protein partners in the regulation of plant tip growth , GA ČR , Martin Potocký
- Funkce rostlinného poutacího komplexu exocyst v exocytóze, buněčném dělení a biogenezi buněčné stěny , GA ČR , Michal Hála
- Úloha diacylglycerolu při toxickém působení hliníku u rostlin , GA ČR , Přemysl Pejchar
- Exocyst, poutací komplex sekretorických váčků, v polarizaci transportu auxinu , GA ČR , Lukáš Synek
- Charakterizace vybraných příslušníků nových skupin rostlinných forminů - třídy II a třídy III. , GA ČR , Viktor Žárský
- The role of the exocyst complex in the plant-pathogen interaction , GA ČR , Tamara Pečenková
- Chracterisation of NADPH oxidase from tobacco pollen and its role in regulation of polar cell expansion , GA ČR , Martin Potocký
- Phosphatidic acid and diacylglycerol-mediated signalling in the polar growth of plant cells , GA AV , Martin Potocký
- Transkriptom mutanta exo70A1 a buněčné funkce EXO70A1, podjednotky komplexu exocyst, u Arabidopsis thaliana , GA AV , Lukáš Synek
Publications
2024
- NATURE CELL BIOLOGY 2024 (External fulltext)
- JOURNAL OF EXPERIMENTAL BOTANY in press 2024 (External fulltext)
- NATURE COMMUNICATIONS 15 2024 (External fulltext)
2023
- FRONTIERS IN PLANT SCIENCE 14 1118670 2023
- PLANT SIGNALLING & BEHAVIOR 18 2163340 2023
- PLANT PHYSIOLOGY 2023 (External fulltext)
2022
- NEW PHYTOLOGIST 233 2185-2202 2022 (External fulltext)
- MOLECULAR PLANT PATHOLOGY 2022 (External fulltext)
- PLANT JOURNAL 2022
- PLANT PHYSIOLOGY 190 1978-1996 2022 (External fulltext)
